Language Язык ภาษา Ngôn ngữ Language Sprache Język Jazyk Limba Nyelv 言語 Língua Idioma Langue Wika Γλώσσα Lingua Valoda Keel Språk Jazyk Мова Språk Language Language
 en
en-
 en
en
-
 de
de
-
 fi
fi
-
 pl
pl
-
 pt
pt
-
 es
es
-
 ro
ro
-
 hu
hu
-
 fr
fr
-
 ph
ph
-
 vn
vn
-
 th
th
-
 cz
cz
-
 no
no
-
 jp
jp
-
 lt
lt
-
 lv
lv
-
 gr
gr
-
 ee
ee
-
 sk
sk
-
 se
se
-
 dk
dk
-
 it
it


Press spin
Нажмите Крутить
Drücken dreh
Paina spin
Naciśnij spin
Pressione spin
Presione Spin
Apăsați Spin
Nyomja meg
a centrifugálást Appuyez sur Spin Pindutin ang Spin Nhấn Spin กดสปิน Stiskněte Spin スピン Trykk på spinn Πατήστε Spin Paspauskite sukimą Nospiediet spin Tryck på spin Vajutage spin Stlačte spin Натисніть Крутити Tryk på spin Premere Spin
a centrifugálást Appuyez sur Spin Pindutin ang Spin Nhấn Spin กดสปิน Stiskněte Spin スピン Trykk på spinn Πατήστε Spin Paspauskite sukimą Nospiediet spin Tryck på spin Vajutage spin Stlačte spin Натисніть Крутити Tryk på spin Premere Spin
Good luck
Удачи
Viel Glück
onnea
powodzenia
boa sorte
Buena suerte
Noroc
sok szerencsét
Bonne chance
Good Luck
Chúc may mắn
โชคดี
Hodně štěstí
幸運を押してください
lykke til
Καλή τύχη
sėkmės
veiksmi
lycka till
palju õnne
veľa šťastia
Удачі
held og lykke
Buona fortuna
SCRATCH THE CARDS
Стереть карты
Kratzen Sie die Karten
Naarmuta kortteja
Zarysuj karty
Arranhe as cartas
Rascar las tarjetas
Zgârie cărțile
Scratch a kártyák
Grattre les cartes
Kiskisan ang mga kard
Cào thẻ
เกาการ์ด
Poškrábat karty
カード
Skrap kortene
Ξύστε τις κάρτες
Nubraižykite kortas
Skrāpēt kartes
Skrapa korten
Kriimustage kaarte
Poškriabajte karty
Стерти картки
Skrabe kortene
Gratta le carte
BIG WIN
большая победа
großen Gewinn
iso voitto
Big Win
grande vitória
gran victoria
câștigul mare
nagy győzelem
grosse victoire
malaking panalo
chiến thắng lớn
ชัยชนะครั้งใหญ่
velké vítězství
大きな勝利を傷つけます
stor gevinst
μεγάλη νίκη
BIG WIN
lielo uzvaru
stor vinst
suur võit
veľkú výhru
Великий виграш
Store sejr
Grande vittoria









600 EUR
600 $
600 NZD
30 000 INR
3000 DKK
600 EUR
600 EUR
2 500 PLN
600 EUR
3 000 BRL
60 000 ARS
425 000 CLP
2 500 PEN
2500000 COP
15 000 MXN
2 400 RON
180 000 HUF
600 CA$
30 000 PHP
15000000 VND
18000 THB
15 000 CZK
60 000¥
6 000 NOK
600 EUR
600 EUR
600 EUR
600 EUR
600 EUR
600 EUR
3000 DKK
600 EUR




600 EUR
600 $
600 NZD
30 000 INR
3000 DKK
600 EUR
600 EUR
2 500 PLN
600 EUR
3 000 BRL
60 000 ARS
425 000 CLP
2 500 PEN
2500000 COP
15 000 MXN
2 400 RON
180 000 HUF
600 CA$
30 000 PHP
15000000 VND
18000 THB
15 000 CZK
60 000¥
6 000 NOK
600 EUR
600 EUR
600 EUR
600 EUR
600 EUR
600 EUR
3000 DKK
600 EUR




600 EUR
600 $
600 NZD
30 000 INR
3000 DKK
600 EUR
600 EUR
2 500 PLN
600 EUR
3 000 BRL
60 000 ARS
425 000 CLP
2 500 PEN
2500000 COP
15 000 MXN
2 400 RON
180 000 HUF
600 CA$
30 000 PHP
15000000 VND
18000 THB
15 000 CZK
60 000¥
6 000 NOK
600 EUR
600 EUR
600 EUR
600 EUR
600 EUR
600 EUR
3000 DKK
600 EUR




600 EUR
600 $
600 NZD
30 000 INR
3000 DKK
600 EUR
600 EUR
2 500 PLN
600 EUR
3 000 BRL
60 000 ARS
425 000 CLP
2 500 PEN
2500000 COP
15 000 MXN
2 400 RON
180 000 HUF
600 CA$
30 000 PHP
15000000 VND
18000 THB
15 000 CZK
60 000¥
6 000 NOK
600 EUR
600 EUR
600 EUR
600 EUR
600 EUR
600 EUR
3000 DKK
600 EUR


YOU WON
ВЫ ВЫИГРАЛИ
SIE HABEN
SINÄ VOITIT
TWOJA WYGRANA TO
VOCÊ GANHOU
GANASTE
AI CASTIGAT
ÖN NYERT
TU AS GAGNÉ
NANALO KA
BẠN ĐÃ THẮNG
คุณได้รับรางวัล
VYHRÁL JSI
あなたが勝った
DU VANT
ΚΕΡΔΙΣΕΣ
TU LAIMĖJAI
TU UZVARĒJI
DU VANN
SINA VÕITSID
VYHRAL SI
ВИ ВИГРАЛИ
DU VANDT
HAI VINTO
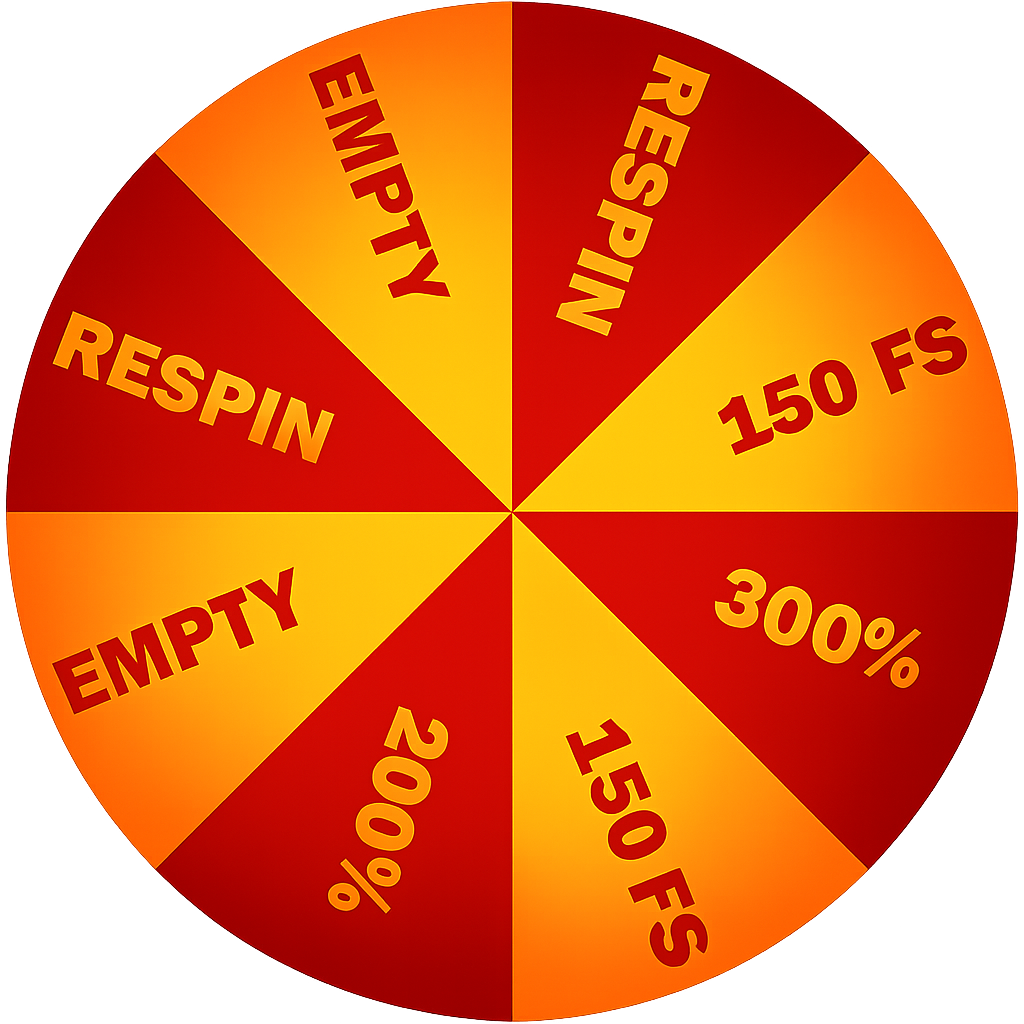
300% BONUS
300% БОНУС
300% BONUS
300% BONUS
300% BONUSU
300% DE BÓNUS
300% DE BONO
300% BONUS
300% BÓNUSZ
300% DE BONUS
300% BONUS
300% TIỀN THƯỞNG
โบนัส 300%
300% BONUS
300% ボーナス
300% BONUS
300% ΜΠΟΝΟΥΣ
300% BONUSAS
300% BONUSS
300% BONUS
300% BOONUS
300% BONUS
300% БОНУС
300% BONUS
300% DI BONUS
PICK UP YOUR WELCOME BONUS
Возьми свой приветственный бонус
HOLEN SIE SICH IHREN WILLKOMMENSBONUS
Nosta tervetuliaisbonus
ODBIERZ SWÓJ BONUS POWITALNY
Pegue seu bônus de boas -vindas
Recoge tu bonificación de bienvenida
Ridicați bonusul dvs. de bun venit
Vedd fel az üdvözlő bónuszát
Procurez-vous votre bonus de bienvenue
Kunin ang iyong maligayang pagdating bonus
Nhận tiền thưởng chào mừng của bạn
รับโบนัสต้อนรับของคุณ
Vyzvedněte si uvítací bonus
ウェルカムボーナス
Hent din velkomstbonus
Πάρτε το μπόνους καλωσορίσματος
Pasiimkite pasveikinimo premiją
Paņemiet savu laipno bonusu
Hämta din välkomstbonus
Võtke oma teretulnud boonus
Zoberte si uvítací bonus
Отримайте свій вітальний бонус
Hent din velkomstbonus
Prendi il tuo bonus di benvenuto
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
300 %
up to 1200 EUR
up to 1200 $
up to 1200 NZD
up to 60 000 INR
up to 6000 DKK
bis zu 1200 €
jopa 1200 euroa
do 5000 PLN
até 1200 EUR
até 6000 BRL
hasta 120 000 ARS
hasta 850000 CLP
hasta 5000 PEN
hasta 5 000 000 COP
hasta 30 000 MXN
până la 4800 RON
akár 360 000 HUF
jusqu'à 1200CA$
hanggang 60 000 PHP
lên đến 30000000 VND
สูงสุด 36 000 THB
až 30 000 CZ
最大 ¥120000 ユーロ
opptil 12 000 NOK
έως 1200 EUR
iki 1200 EUR
līdz 1200 EUR
upp till 1200 euro
kuni 1200 eurot
až 1200 EUR
op til 6000 DKK
fino a 1200 EUR
Bonus T&C's
Правила и условия
Bonus T&Cs
Bonus T&C:t
Bonus T&C
Bônus T&C's
Bonus T&C's
Bonus T&C
Bónusz T&C
Bonus T&C
Bonus T&C's
Tiền thưởng T & C
โบนัส T&C
Bonus T&C
ボーナスT&C
Μπόνους T & C's
Premija T&C
Bonuss t & c
Bonusvillkor
Boonus T & C -d
Bonus T&C
Правила та умови
Bonus T & C-er
Bonus T & C'er
Bonus T&C's